Saxitoxin
Background
Saxitoxins (mytilotoxin; shellfish toxins; STX, PSP) are neurotoxic poisons produced naturally by certain types of planktonic algae (dinoflagellates) (Wiese et al., 2010). The consumption of shellfish (e.g. mussels, butter clams and oysters) that have accumulated the toxin when feeding on these organisms may cause paralytic shellfish poisoning in humans (mytilotoxism). It should be noted that the concentration of the toxin can be higher still - as much as 10 mg of saxitoxin per 100 g of mussel flesh – when algal blooms occur. Another source of PSP toxins are freshwater cyanobacteria (e.g. Aphanizomenon flos-aquae). The first complete chemical analysis and synthesis of saxitoxin was conducted by Kishi in 1977. Given that it is extremely difficult to synthesise, saxitoxin (and its derivatives) cannot be readily procured.
Saxitoxin as a bioweapon
Classified as Agent TZ (chemical weapon designation), saxitoxin is considered to have chemical weapon potential due to its high toxicity. Researchers have shown that it would be possible to spike ammunition with saxitoxin, thus ensuring the immediate death of the victim. Saxitoxin is much more poisonous than the synthetic nerve gas sarin and, like the phytotoxin ricin, is also classified as a biological weapon. Saxitoxin is listed in the Chemical Weapons Convention (Schedule 1). The toxin also appears in the Annex (War Weapons List) to the German War Weapons Control Act.
Chemical structure and properties
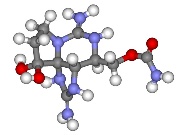
The best known PSP toxin is saxitoxin. Further 20 PSP toxins which have been isolated to date, show a high structural similarity to saxitoxin. PSP toxins are soluble in water, methanol and ethanol, but not in other organic solvents. They remain stable in slightly acidic environments, but tend to oxidise when exposed to alkaline conditions.
Toxicity
Saxitoxin (STX) is a powerful neurotoxin. Like tetrodotoxin, STX binds to the sodium channels in nerve cells, thereby blocking the influx of sodium ions (Fig. 2). A single STX molecule is enough to block one sodium channel. The two guanidine sub-structures present in saxitoxin must be protonated. The toxicity of saxitoxin is much greater than that of synthetic neurotoxins. Consequently, it ranks alongside other toxins such as ricin and botulinum neurotoxin as one of the most poisonous chemical substances known to mankind. LD50 (mouse): oral: 263 µg/kg, intraperitoneal: 10 µg/kg (van Apeldoorn et al., 2007).
Analysis
A variety of biological, biochemical and chemical procedures can be used to analyse saxitoxin (Humpage et al., 2010). These can be divided into two groups: non-chromatographic and chromatographic. The first group includes bioassays, immunoassays and fluorimetric assays. However, these are only capable of producing a summary identification of several PSP toxins (total toxicity). However, over the last 15 years, highly sensitive chromatographic procedures have been developed which make it possible to identify individual compounds with greater precision. Over time, the HPLC method, which uses fluorescence detection, has become the standard procedure (DIN EN 14526).
References
Wiese M, D'Agostino PM, Mihali TK, Moffitt MC, Neilan BA. Neurotoxic alkaloids: saxitoxin and its analogs. Mar Drugs. 2010 Jul 20;8(7):2185-211. Review.
van Apeldoorn ME, van Egmond HP, Speijers GJ, Bakker GJ. Toxins of cyanobacteria. Mol Nutr Food Res. 2007 Jan;51(1):7-60. Review.
Humpage AR, Magalhaes VF, Froscio SM. Comparison of analytical tools and biological assays for detection of paralytic shellfish poisoning toxins. Anal Bioanal Chem. 2010 Jul;397(5):1655-71. Epub 2010 Jan 26. Review.
van Apeldoorn ME, van Egmond HP, Speijers GJ, Bakker GJ. Toxins of cyanobacteria. Mol Nutr Food Res. 2007 Jan;51(1):7-60. Review.
Humpage AR, Magalhaes VF, Froscio SM. Comparison of analytical tools and biological assays for detection of paralytic shellfish poisoning toxins. Anal Bioanal Chem. 2010 Jul;397(5):1655-71. Epub 2010 Jan 26. Review.
WHO guidance: http://www.who.int/csr/delibepidemics/annex2.pdf
DIN EN 14526: Foodstuffs - Determination of saxitoxin and dc-saxitoxin in mussels - HPLC method using pre-column derivatization with peroxide or periodate oxidation; German version, 2004.